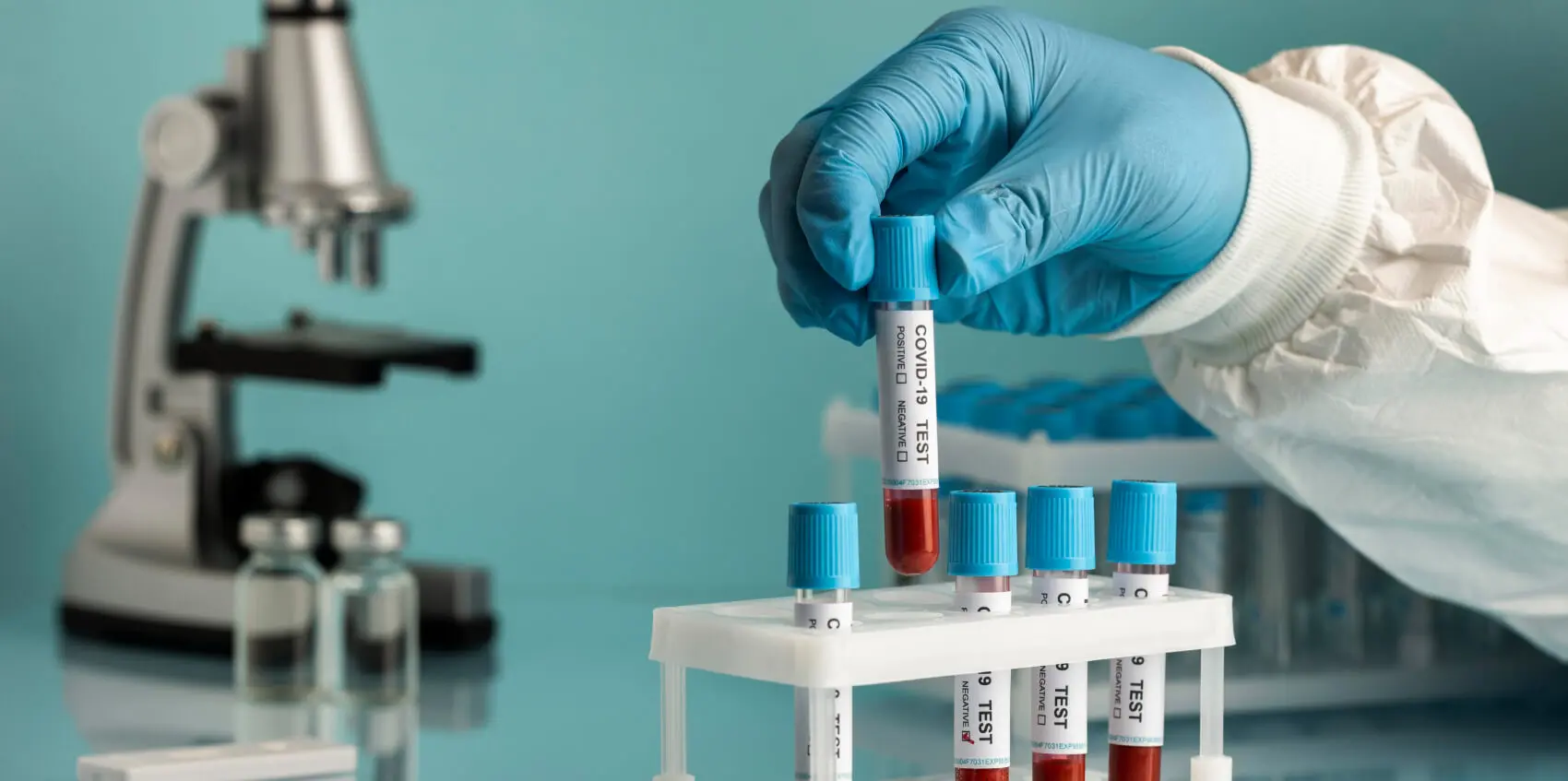
Hematology
Clinical Chemistry
Microbiology
Immunology and Serology
Cytology and Histology
Blood Specimens
Clean Catch Urine
Time-Sensitive Specimens
Women’s Health and Prenatal Testing
Molecular Diagnostics
Immunology and Serology
Cytology and Histology
Blood Specimens
Clean Catch Urine
Time-Sensitive Specimens
Women’s Health and Prenatal Testing
Plot 66, BLK F Ejisu Asamang, Ashanti, Ghan
Downloads
Download Profile.PDF
Download Profile.DOC
Book This Services
cGMP-Compliant Infrastructure
Our physical facility is a "befitting edifice" designed specifically to meet the highest international standards of pharmaceutical manufacturing

A Facility Designed for Excellence
The Aspee plant in Ejisu-Asamang is a marvel of modern industrial design. Every square inch of the facility was planned to facilitate a smooth, logical flow of materials and personnel, which is a core requirement of Good Manufacturing Practices. By separating raw material handling, processing, and packaging, we eliminate the risk of cross-contamination—a critical factor in pharmaceutical safety.
- Purpose-Built Controlled Environment: Our befitting edifice is a carefully engineered facility designed as a controlled manufacturing environment, not merely a physical structure.
- Integrated Environmental Control Systems: HEPA filtration, precise climate control, and specialized lighting operate in unison to maintain optimal conditions for drug stability and integrity.
- Capability for Sensitive Formulations: This advanced infrastructure enables us to safely and consistently handle delicate and complex formulations that many facilities cannot support.
- Premium Regional Manufacturing Position: By investing in world-class infrastructure, Aspee is positioned as a premium pharmaceutical manufacturer within the West African sub-region.
Modern Machinery and Technical Superiority:
Inside our process rooms, you will find the best cGMP-compliant machinery available. We invest heavily in technology that automates repetitive tasks, ensuring a level of consistency that manual processes simply cannot match. From high-speed tablet presses to precision liquid filling machines, our equipment is chosen for its reliability and its ability to maintain the integrity of our formulations.
- High-Volume Versatile Manufacturing: Our advanced technical capabilities enable large-scale production while supporting a wide range of pharmaceutical manufacturing needs.
- Scalable Infrastructure for Growing Demand: We are equipped to expand output seamlessly, ensuring we can meet the increasing healthcare needs of the Ghanaian population.
- Uncompromised Attention to Quality: Even at high production volumes, we maintain strict control over the fine details that preserve product safety, consistency, and efficacy.
- Internationally Competitive Facility Standards: Our manufacturing environment meets standards comparable to those in more developed pharmaceutical markets, earning confidence from international partners.

Why Choose Us
World-Class cGMP Manufacturing Technology
We operate with best-in-class, cGMP-compliant machinery that automates critical processes, delivering unmatched consistency, precision, and formulation integrity across every production run.
Scalable, High-Volume Manufacturing Capability
Our advanced and versatile infrastructure is designed for scale, allowing us to meet increasing demand without compromising the fine controls that safeguard product quality.
International-Standard Production Environment
With infrastructure and processes that rival facilities in developed pharmaceutical markets, we offer partners the confidence of manufacturing in a truly world-class environment.
"The strength of the medicine begins with the integrity of the walls within which it is created."
We are located in Ejisu-Asamang, near Kumasi, in the Ashanti Region of Ghana.
We host scheduled tours for institutional partners, regulators, and academic groups.
